 Now between F and F-: F- has one more electron, which causes the radius to be bigger. 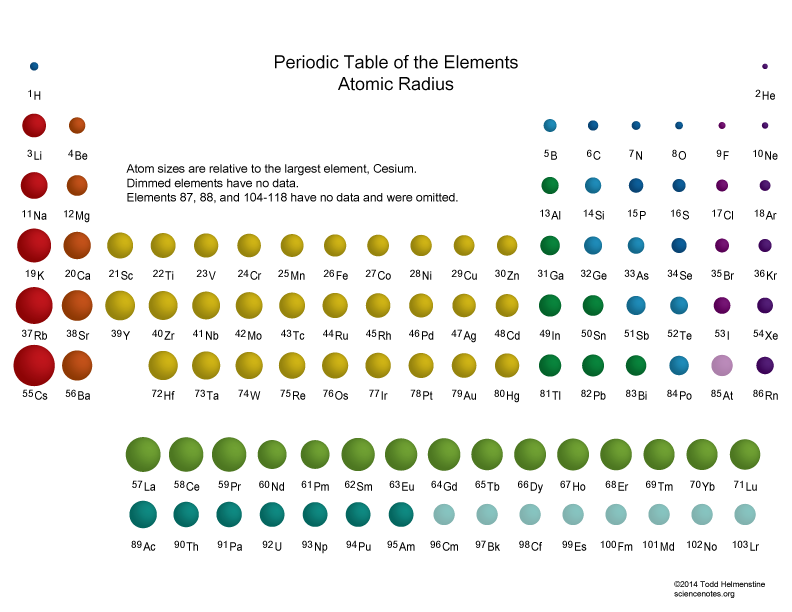
We know that F and F- are both smaller than the rest because they have less shells. But since Te has one shell more than Rb+ is it bigger. Compare electron affinities and electronegativities. We know that Te is smaller that Rb because of the trend of decreasing radius to the right within a period. Predict greater or smaller atomic size and radial distribution in neutral atoms and ions. Another reasoning is that since Rb+ has the same number of protons but less electrons, the electrons are held more tightly in Rb+. We know that Rb+ is smaller than Rb because it loses an electron and has one less shell. Rb and Te are in period 5, and F is in period 2. Atomic Radius of the elements Hydrogen, 53 pm, Niobium, 198 pm, Thallium Helium, 31 pm, Molybdenum, 190 pm, Lead Lithium, 167 pm, Technetium, 183 pm, Bismuth. List the following species in order of increasing size: Rb, Rb+, F, F-, Te.įirst look in which periods the element is located. K+ and Ca 2+įor isoelectronic cations, the more positive the ionic charge, the smaller the ionic radius.įor isoelectronic anions, the more negative the ionic charge, the larger the ionic radius. Size of the Elements on the Periodic Table Periodic table showing the relative sizes of the elements based on atomic radius data.  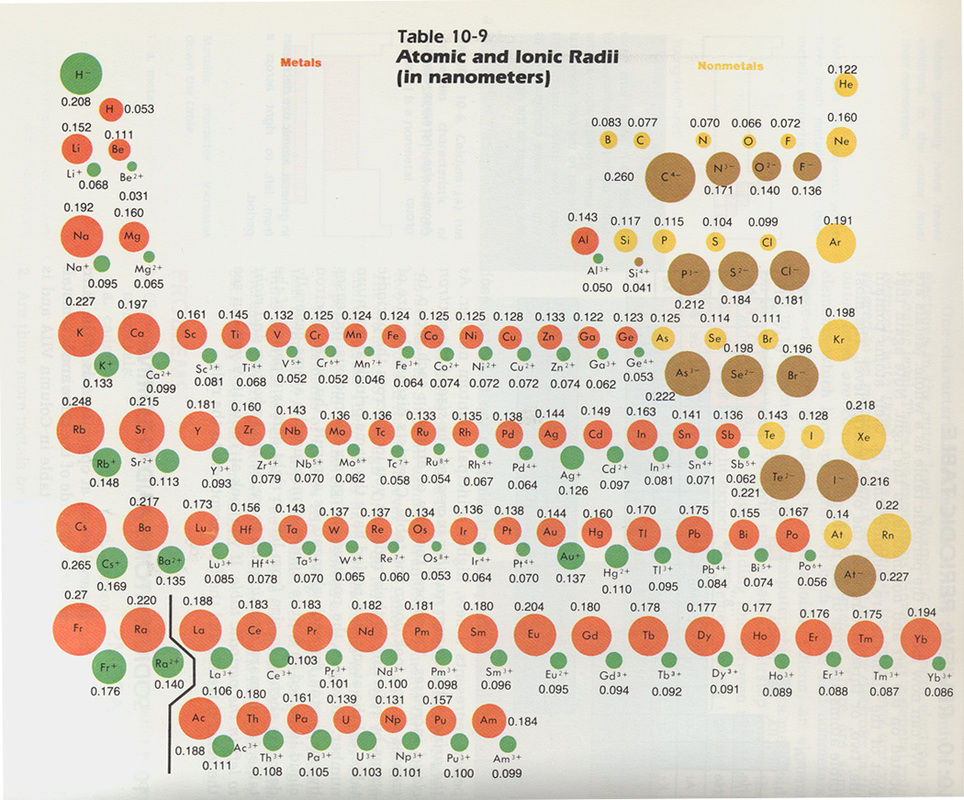
In both cases, there is a periodic table trend. This creates a larger negative charge in the electron cloud than positive charge in the nucleus, causing the electron cloud to 'puff out' a little bit as an ion.Ĭations (or anions) of different elements are said to be **isoelectronic** if they both have the same electronic configuration e.g. The atomic radius of an element is a measure of the size of its atoms. Updated on NovemThe size of atoms of elements may be expressed in terms of atomic radius or ionic radius. The trends for the entire periodic table can be seen in Figure 4.3.1. This trend is illustrated for the covalent radii of the halogens in Table 4.3.1 and Figure 4.3.1. Q1: Why does the size of the elements increase on moving down the groups in a Periodic Table Answer: This is because there takes place an addition of energy levels as we move down in a group in the Periodic Table.As the number of energy levels increases, the size of the atoms also increases. Why? Generally, non-metals gain electrons to achieve the octet. Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. Atomic Radius in Periodic Table Questions with Solutions. Non-metals - the atomic radius of a non-metal is generally smaller than the ionic radius of the same element. This creates a larger positive charge in the nucleus than the negative charge in the electron cloud, causing the electron cloud to be drawn a little closer to the nucleus as an ion. Why? Generally, metals loose electrons to achieve the octet. Metals - the atomic radius of a metal is generally larger than the ionic radius of the same element. This article is part of the ChemHelp Tutoring Wiki 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
